Van Der Waals Equation Example
Van der Waals equation is related to the general gas equation PV=nRT. Both equations are for unlike natures of gases. Van der Waals equation is for Real gases and the platonic gas equation is for ideal gases. This equation was given past Johannes Diederick van der Waals in 1873.
This equation is very helpful to explicate or state the physical country of a non-ideal gas or existent gas. Van der Waals equation has a contrast with the volume, molecular size as well equally molecular interaction of ideal gas. To empathize these two equations, we have to understand the basic divergence between ideal gas and real gas.
Difference between Existent and Ideal gas
A real gas is also referred to as non-platonic gas. And then, the basic difference between these two gases is as under:
| Real GAS | Platonic GAS |
| 1. Information technology does not follow the real gas equation. | one. Information technology follows the ideal gas equation. |
| 2. At low force per unit area and high-temperature Real gas will follow the platonic gas equation. | 2. At all the temperature and pressure level, it follows the platonic gas equation…… |
| 3. In Real gas, molecules are rigid and spheres have the same volume. | 3. Molecules of gas are point masses having negligible volume. |
| 4. There exists a force of attraction betwixt molecules due to their large size. | 4. There exists no Forcefulness of attraction between molecules due to their small size. |
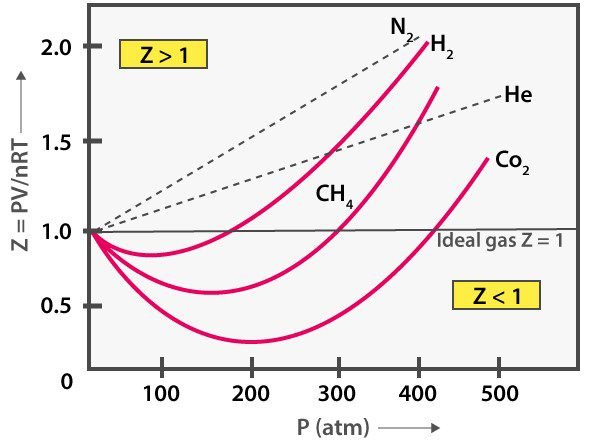
Compressibility gene "Z"
The compressibility factor is denoted by Z. It is the ratio between the Volume of real gas to the volume of an platonic gas.
Z = Fivereal gas / Videal gas
- It also gives an idea nearly the positive, negative, and aught deviation between Vreal gas and Videal gas.
- It also tells at which force per unit area gas beaves as ideal or real gas. If compressibility is equal to one then it volition be Ideal gas but if it is greater or less than one and so it will be a real gas.
- Z = PV/RT; if Z = ane ⇒ Ideal gas merely if Z ≠ ane ⇒Real gas
- In real gases, two possibilities take place Z > i or Z < 1.
- If Z is greater than 1 then the volume of real gas dominates over the book of an ideal gas which causes or enhances the gas to expand and repulsive forces dominate to follow the +ve deviation.
- But if Z is less than one so there is a vice-versa situation of the in a higher place explained. This means Videal > Vreal which causes gas to shrink and to boss attractive forces which causes -ve deviation.
- In +ve or -ve deviation measured volume has a departure from the expected book.
Van der Waals Equation & Derivation
Van der Waals equation gives the relationship between the corrected volume and corrected pressure. It too shows an interesting human relationship between temperature and the amount of gas. The existent gas equation is as well chosen the Van der Waals equation. This equation can exist written every bit:
(P – an2/V2)(Five – nb) = nRT
Whereas,
- P, V, & T are Force per unit area, Volume, and temperature.
- anii/Five2 ⇒ Pressure correction.
- nb ⇒ Volume correction.
- a, b ⇒ Van der Waals constants.
- north ⇒ No. of moles of gas.
Derivation of Van der Waals Equation
This equation will follow the Not-ideal gas. This equation is basically based upon correction in volume and pressure level. If we consider Ideal gas they follow Kinetic molecular theory and the Kinetic equation of gases. An platonic gas is basically based on iii postulates of KMT.
- The volume of molecules is negligible as compared to the book of the container. (Basically, the volume of gas is available space in a container in which it can move freely).
- Equally they are considered betoken masses so didn't possess the force of attraction between them.
- All the collisions between the molecules of gas are perfectly rubberband.
But if we talk about not-ideal gas, they show many deviations from the ideal gas. Van der Waals equation is based on bones ii postulates:
- If we talk well-nigh volume then molecules of real gas are greater which did non occupy a small volume. And then, the book cistron is affected and it is necessary to correct it.
- Due to the large molecular size of the gas, in that location is a greater strength of attraction which causes correct gene pressure level.
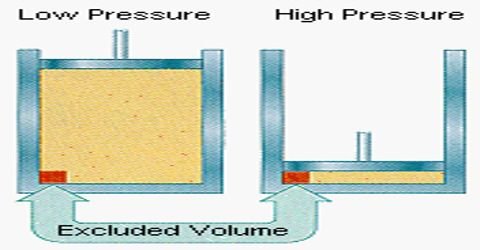
Volume Correction
As it is discussed earlier the cause of volume correction is molecular size. Then,
The book of gas = Vcontainer – Vmolecules
Vg = V – b
b is the excluded book. So, for n number of moles. b is likewise the van der Waals constant and its units are liter per mole .
Vg = V – nb
If we find the total book and so past using the formula:
Total volume = No. of particles × Volume of unmarried-molecule
Vg = n × 4/3πr3
This equation occurs where molecules are independent or complimentary to move but in the Existent gas case, molecules have the force of allure betwixt them which did not allow whatever independent motion and have interaction with each other.
If two molecules are interacting and then volume correction becomes:
b = north × four/3πrthree × two
b = 8/three north π r3
For a unmarried molecule it becomes;
b = 4 × 4/3 n π r3
b = 4n × Volume of a single molecule of gas
For due north number of moles;
nb = 4n × Volume
So, Book = Five – nb
A very noticeable cistron in book correction is the significance of b. b is directly proportional to the size of a molecule of gas.
b ∝ Size of a molecule of gas
Pressure Correction
In the case of pressure where there is an ideal gas. These gas molecules strike on the walls of the container and perform elastic collisions which exert Pressure "P" on the walls of the container due to no Force of allure.
Only in real gas, there is an exception. No molecular collisions or wall collisions take place due to their greater size and they didn't strike too hard. So, in that location is a reduction in the value of force per unit area.
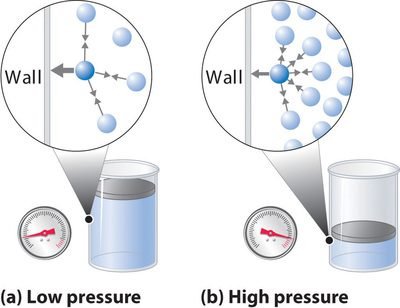
So, the total force per unit area of a gas is the sum of force per unit area and the internal pressure between particles.
Pgas = P + Pinternal
Every bit there are 2 unlike molecules of gas with large molecular sizes A & B. And then,
Concentration of A-blazon molecule = northward/V
Concentration of B-blazon molecule = n/5
Concentration is basically molarity and molarity is defined as the number of moles per unit volume. So,
Pinternal ∝ (A)(B)
Pinternal ∝ (n/V)(northward/Five)
Pinternal = an2/Fiveii
a is van der Waals abiding. Every bit nosotros know,
Pgas = P + Pinternal
Pgas = P + antwo/Vii
The significance of a is essential is pressure correction. a is directly proportional to the forcefulness of allure and tooth mass. An increment in "a" causes an increase in the intermolecular force of attraction which causes the liquefaction of gases.
Just it is important to notice that the small size atom of a gas molecule has zero forcefulness of attraction. So, their pressure correction constant is equal to zero.
If we substitute the original pressure and volume with pressure correction and volume correction and so the general gas equation becomes:
PV = nRT
(P – antwo/Five2)(Five – nb) = nRT
How Van der Waals equation is related to compressibility cistron Z?
By using this equation, we will know a human relationship between the compressibility factor Z .
- If the temperature is abiding then an increment in volume causes a decrease in pressure. So, correction in book is negligible as compared to actual volume. So, Z < i which gives a negative deviation, and Vreal < Vplatonic. In this case, attractive forces dominate.
- If the temperature is constant then an increase in pressure causes a decrease in book. So, the correction in pressure is negligible every bit compared to the bodily pressure applied. And then, Z > one which gives a positive deviation, and 5real > Fiveideal. In this instance, repulsive forces boss.
Uses of Van der Waals equation
- It allows the prediction of gases betters than the ideal gas equation.
- Book correction and pressure correction terms are not but for gases but as well for fluids.
- It has the ability to calculate some of the critical conditions at which the liquefication of gas occurs.
- The results derived from the Van der Waals equation have accuracy below the critical temperature.
- At high pressure, it allows for the creation of an approximation for existent gases and likewise allows the prediction of a non-ideal gas.
Van Der Waals Equation Example,
Source: https://www.uochemists.com/van-der-waals-equation/
Posted by: cookplive1938.blogspot.com


0 Response to "Van Der Waals Equation Example"
Post a Comment